The Importance of Validated Medical Devices
In clinical settings, validated medical devices are essential because they enable reliable patient diagnosis, care, and monitoring. These devices have undergone extensive testing and validation to guarantee that they are secure, dependable, and accurate.
The confidence that readings or measurements are reliable and consistent is one of the key advantages of using validated medical devices. When it comes to identifying and managing chronic diseases like diabetes, hypertension, and heart disease, this is especially crucial. For healthcare providers to make educated judgments about patient care and to modify treatment plans as necessary, accurate measurements are crucial. Another benefit of using validated medical devices is that they can reduce the risk of errors or misinterpretation of results. For example, if a home blood pressure is not validated, it may provide inaccurate readings that can lead to incorrect therapeutic interventions, which can be dangerous for patients.
VDL and Blood Pressure Monitors
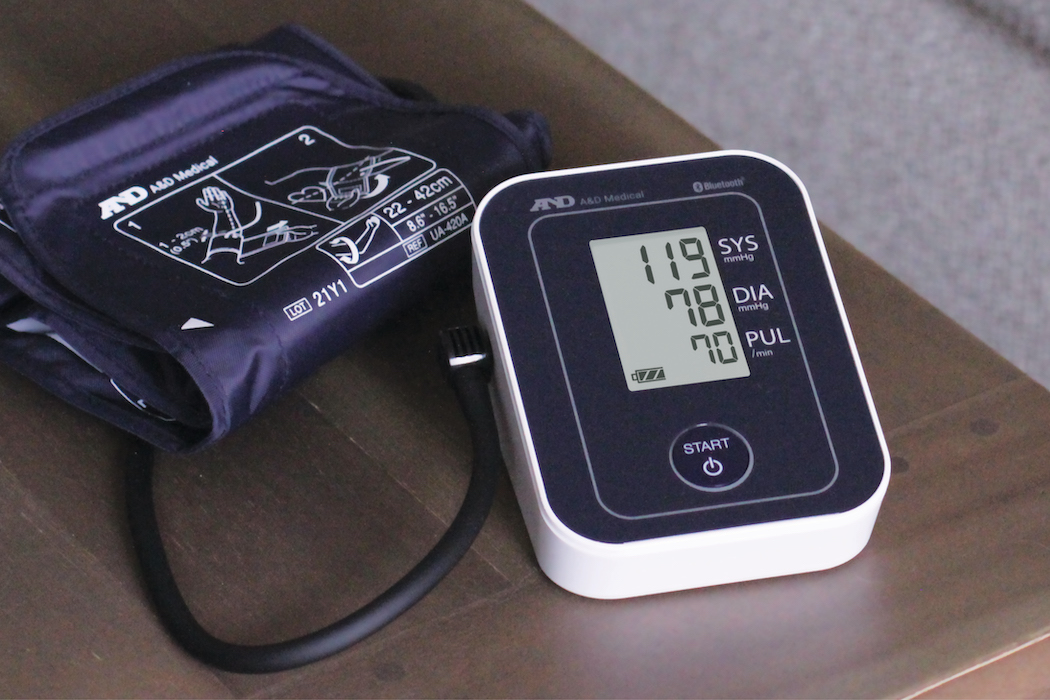
There are many different types of blood pressure monitors available on the market, including manual sphygmomanometers, home blood pressure monitors, professional office blood pressure monitors, blood pressure kiosks, and ambulatory blood pressure monitors. However, not all these devices have been validated to ensure their accuracy and reliability. A recent study by Dr. Dean Picone from the University of Tasmania, Australia found that over 75% of blood pressure monitors are not validated.
The main cause of death and disability is uncontrolled high blood pressure (“BP”). For the proper diagnosis and treatment of hypertension, reliable blood pressure measurement is crucial. Whether or not the BP measurement instrument has undergone clinical accuracy validation is one critical factor in accurate measurement.
The American Medical Association (AMA) and NORC are trying to change how clinicians and consumers can reliably determine if a blood pressure monitor is validated. The AMA enlisted the National Opinion Research Center at the University of Chicago (NORC) to help in the design and management of an impartial process to identify which BP devices offered in the U.S. meet the AMA’s established criteria to validate clinical accuracy (the Validated Device Listing Criteria). This was done to address the growing issue of unvalidated devices being marketed in the US. A BP device’s compliance with the VDL Criteria for validation of clinical accuracy is determined by an independent review committee made up of doctors with advanced training in the field of blood pressure. The “US Blood Pressure Validated Device Listing” or “VDL” is a formal list of BP devices that have undergone clinical accuracy validation because of this independent evaluation procedure.
For healthcare providers, the VDL is a reference to monitors that have proven accuracy when choosing a blood pressure monitor for their office, like Automated Office Blood Pressure (AOBP), kiosks or Ambulatory Blood Pressure Monitors (ABPM) or even recommending home monitors to patients.
The Validated Device List has listed A&D Medical devices on their website. This includes our best-selling blood pressure monitors. Below is a list of our approved devices that you can purchase for your clinic or home use: